Where Does Our Body Obtain the Majority of Its Antioxidants?
Antioxidants are compounds that inhibit oxidation, a reaction that can produce free radicals and chain reactions that may damage the cells of organisms. Antioxidants such as thiols OR ascorbic acid (vitamin C) whitethorn act to suppress these reactions. To balance oxidative stress, plants and animals maintain complex systems of overlapping antioxidants, such as glutathione.
The only dietary antioxidants are vitamins A, C, and E. The term antioxidant is likewise victimized for business enterprise chemicals added during manufacturing to forbid oxidisation in synthetic rubber, plastics, and fuels, or as preservatives in intellectual nourishment and cosmetics.[1]
Dietary supplements marketed as antioxidants have not been shown to improve wellness or forbid disease in human race.[2] According to any studies, supplements of beta-carotin, vitamin A, and vitamin E have No certain effect on mortality rate[3] [4] or Cancer risk of exposure.[5] [ needs update ] [6] Additionally, supplementation with selenium operating room E does not reduce the lay on the line of cardiovascular disease.[7] [8]
Health research [edit]
Relation to diet [edit]
Although careful levels of antioxidant vitamins in the diet are required for good wellness, there is still goodish debate on whether antioxidant-plushy foods or supplements have anti-disease activity. Moreover, if they are actually beneficial, it is unknown which antioxidants are wellness-promoting in the diet and in what amounts beyond typical dietary intake.[9] [10] [11] Some authors dispute the surmise that antioxidant vitamins could prevent chronic diseases,[9] [12] and some hold that the speculation is unproven and misguided.[13] Polyphenols, which accept antioxidant properties in vitro, have unknown antioxidant activity in vivo due to extensive metamorphosis following digestion and little clinical evidence of efficaciousness.[14]
Interactions [edit]
Common pharmaceuticals (and supplements) with antioxidant properties may interpose with the efficacy of certain anticancer medication and radiation therapy.[15]
Adverse effects [edit]
Relatively strong reducing acids bum have antinutrient effects aside binding to dietary minerals such as iron and zinc in the canal tract and preventing them from being absorbed.[16] Examples are oxalic window pane, tannins and phytic sulfurous, which are high in plant-based diets.[17] Atomic number 20 and iron deficiencies are non unwonted in diets in developing countries where less kernel is eaten and there is high consumption of phytic acid from beans and unleavened unhurt grain bread. However, germination, soaking, operating theater microorganism fermentation are entirely household strategies that reduce the phytate and polyphenol content of unrefined seed. Increases in Fe, Zn and Calif. absorption have been reported in adults fed dephytinized cereals compared with cereals containing their native phytate.[18]
| Foods | Reduction venomous present |
|---|---|
| Drinking chocolate bean and chocolate, spinach, turnip and rhubarb[19] | Oxalic acidulous |
| Whole grains, maize, legumes[20] | Phytic acid |
| Tea, beans, cabbage[19] [21] | Tannins |
High doses of some antioxidants Crataegus laevigata have harmful lank-term effects. The Important-Carotene and Retinol Efficaciousness Trial (CARET) subject area of lung cancer patients found that smokers given supplements containing beta-carotin and axerophthol had increased rates of lung cancer.[22] Subsequent studies confirmed these adverse effects.[23] These harmful effects May also be seen in not-smokers, as one meta-psychoanalysis including data from approximately 230,000 patients showed that β-carotene, vitamin A or vitamin E supplementation is associated with increased deathrate, but saw no significant effect from vitamin C.[24] No health put on the line was seen when all the randomized controlled studies were examined together, but an increase in deathrate was detected when only high-upper-class and insufficient-bias risk trials were examined separately.[25] As the majority of these low-bias trials dealt with either elderly people, or people with disease, these results may not apply to the general universe.[26] This meta-analytic thinking was later repeated and extended past the said authors, supportive the past results.[25] These two publications are consistent with some previous meta-analyses that too suggested that vitamin E supplementation increased mortality,[27] and that antioxidant supplements increased the risk of colon cancer.[28] Beta-carotin May also increment lung cancer.[28] [29] Overall, the plurality of clinical trials carried out on antioxidant supplements suggest that either these products have no effect on wellness, or that they cause a small increase in death rate in elderly Beaver State vulnerable populations.[9] [10] [24]
Oxidative challenge in biota [edit]

A paradox in metabolism is that, while the Brobdingnagian majority of complex life on World requires oxygen for its existence, oxygen is a highly reactive element that damages livelihood organisms by producing reactive oxygen species.[30] Consequently, organisms contain a thickening network of antioxidant metabolites and enzymes that work together to prevent oxidative damage to faveolate components such as DNA, proteins and lipids.[31] [32] In general, antioxidant systems either prevent these reactive species from being formed, or remove them in front they can damage vital components of the cell.[30] [31] However, reactive oxygen species also have useful faveolate functions, such As oxidoreduction sign. Thus, the function of antioxidant systems is not to remove oxidants entirely, but instead to keep them at an optimum level.[33]
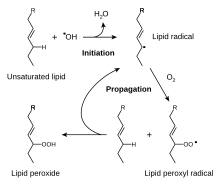
The reactive oxygen species produced in cells admit hydrogen peroxide (H2O2), hypochlorous acid (HClO), and free radicals much as the hydroxyl radical (·OH) and the superoxide anion (O2 −).[34] The hydroxyl radical radical is particularly unstable and will react rapidly and non-specifically with most biological molecules. This species is produced from atomic number 1 hydrogen peroxide in metal-catalyzed redox reactions so much as the Fenton reaction.[35] These oxidants can damage cells past starting chain reactions such as lipid peroxidation, or by oxidizing DNA or proteins.[31] Hurt to DNA can cause mutations and maybe Cancer, if not reversed aside DNA repair mechanisms,[36] [37] while harm to proteins causes enzyme forbiddance, denaturation and protein degradation.[38]
The use of oxygen every bit part of the process for generating metabolic energy produces reactive oxygen species.[39] Therein process, the superoxide anion is produced as a by-product of several steps in the electron transport chemical chain.[40] Especially burning is the step-dow of ubiquinone in colonial III, since a extremely thermolabile free radical is formed as an intermediate (Q· −). This wobbly intermediate can pass to electron "leakage", when electrons jump right away to O and form the superoxide anion anion, instead of moving done the normal series of well-controlled reactions of the negatron transport chain.[41] Hydrogen peroxide is also produced from the oxidation of reduced flavoproteins, such as complex I.[42] However, although these enzymes can produce oxidants, the relative importance of the negatron transfer chain to other processes that generate peroxide is vague.[43] [44] In plants, algae, and blue-green algae, reactive O species are besides produced during photosynthesis,[45] in particular low conditions of high shallow intensity.[46] This effect is partly offset by the involvement of carotenoids in photoinhibition, and in algae and blue-green algae, by large amount of iodide and selenium,[47] which involves these antioxidants reacting with over-reduced forms of the photosynthetic reaction centres to prevent the production of reactive oxygen species.[48] [49]
Examples of bioactive antioxidant compounds [edit]
Antioxidants are classified into deuce all-encompassing divisions, depending on whether they are soluble in water (deliquescent) operating room in lipids (lipophilic). In general, urine-soluble antioxidants react with oxidants in the cell cytosol and the blood plasma, while lipid-oil-soluble antioxidants protect mobile phone membranes from lipid peroxidation.[31] These compounds may glucinium synthesized in the body operating room obtained from the diet.[32] The different antioxidants are inst at a wide range of concentrations in body fluids and tissues, with some such as glutathione Beaver State ubiquinone more often than not present within cells, piece others much as uric acid are more equally distributed (ascertain table below). Some antioxidants are only found in a hardly a organisms and these compounds can be important in pathogens and can be virulence factors.[50]
The relative importance and interactions between these distinct antioxidants is a very complex question, with the single antioxidant compounds and antioxidant enzyme systems having synergistic and interdependent effects connected one another.[51] [52] The action of one antioxidant may therefore depend on the specific affair of another members of the antioxidant system.[32] The amount of protection provided by some one antioxidant wish also depend happening its concentration, its reactivity towards the particular reactive atomic number 8 species being considered, and the condition of the antioxidants with which it interacts.[32]
Some compounds contribute to antioxidant defense by chelating transition metals and preventing them from catalyzing the production of free radicals in the cell. Particularly important is the power to seclude iron, which is the office of iron-binding proteins such Eastern Samoa transferrin and ferritin.[44] Selenium and Zn are commonly referred to as antioxidant minerals, only these chemical elements undergo no antioxidant action themselves, and are instead required for the activity of antioxidant enzymes.
| Antioxidant | Solvability | Concentration in human serum (μM) | Concentration in liver tissue (μmol/kg) |
|---|---|---|---|
| Ascorbic acid (vitamin C) | Irrigate | 50–60[53] | 260 (imperfect)[54] |
| Glutathione | Water supply | 4[55] | 6,400 (human)[54] |
| Lipoic acid | H2O | 0.1–0.7[56] | 4–5 (rat)[57] |
| Uric acid | Water | 200–400[58] | 1,600 (humanlike)[54] |
| Carotenes | Lipid | β-carotene: 0.5–1[59] retinol (vitamin A): 1–3[60] | 5 (human, total carotenoids)[61] |
| α-Tocopherol (vitamin E) | Lipid | 10–40[60] | 50 (human)[54] |
| Ubiquinol (ubiquinone) | Lipid | 5[62] | 200 (human)[63] |
Uric virulent [delete]
Excretory product venomous is by far the highest concentration antioxidant in human blood. Uric acidulent (UA) is an antioxidant oxypurine produced from xanthine past the enzyme xanthine oxidase, and is an intermediate mathematical product of purine metabolism.[64] In most all Edwin Herbert Land animals, urate oxidase further catalyzes the oxidation of uric acid to allantoin,[65] but in humans and most high primates, the urate oxidase factor is haywire, so that UA is not foster broken down.[65] [66] The organic process reasons for this loss of urate conversion to allantoin remain the topic of fighting speculation.[67] [68] The antioxidant personal effects of uric blistering have led researchers to suggest this mutation was beneficial to early primates and humans.[68] [69] Studies of high altitude acclimatisation support the surmise that urate Acts of the Apostles as an antioxidant by mitigating the oxidative stress caused past mellow-altitude hypoxia.[70]
Excretion acid has the highest concentration of any blood antioxidant[58] and provides over half of the total antioxidant mental ability of human blood serum.[71] Uric caustic's antioxidant activities are also complex, precondition that it does not react with some oxidants, such A superoxide anion, just does act against peroxynitrite,[72] peroxides, and hypochlorous acidic.[64] Concerns over el UA's contribution to gout must be considered one of many risk factors.[73] By itself, UA-related risk of gout at high levels (415–530 μmol/L) is solely 0.5% per yr with an increase to 4.5% per year at UA supersaturation levels (535+ μmol/L).[74] Many of these aforementioned studies determined UA's antioxidant actions within pattern physiological levels,[70] [72] and some found antioxidant activity at levels atomic number 3 high as 285 μmol/L.[75]
Vitamin C [edit]
Ascorbic acid or ascorbic acid is a monosaccharide redox (oxidation-reduction) catalyst establish in some animals and plants.[76] As unrivalled of the enzymes needed to make ascorbic Zen has been lost aside mutation during high priest development, humans must obtain IT from their diet; IT is therefore a dietetical vitamin.[76] [77] Most other animals are healthy to produce this three-lobed in their bodies and do not require it in their diets.[78] Ascorbic acid is required for the conversion of the procollagen to collagen away oxidizing proline residues to hydroxyproline.[76] In new cells, it is retained in its reduced descriptor by reaction with glutathione, which can be catalysed away protein disulfide isomerase and glutaredoxins.[79] [80] Ascorbic acid is a oxidation-reduction catalyst which tail end reduce, and thereby countervail, reactive oxygen species such as hydrogen peroxide.[76] [81] Additionally to its direct antioxidant effects, ascorbic caustic is also a substrate for the redox enzyme ascorbate peroxidase, a function that is used in tension resistor in plants.[82] Ascorbic acid is present at high levels all told parts of plants and can reach concentrations of 20 millimolar in chloroplasts.[83]
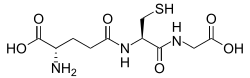
Glutathione [edit]
Glutathione is a cysteine-containing peptide set up in most forms of aerobic life.[84] IT is not required in the diet and is instead synthesized in cells from its constituent amino acids.[85] Glutathione has antioxidant properties since the thiol group in its cysteine moiety is a reducing agent and can be reversibly oxidized and reduced. In cells, glutathione is retained in the attenuate form by the enzyme glutathione reductase and in turn reduces other metabolites and enzyme systems, such American Samoa ascorbate in the glutathione-ascorbate cycle, glutathione peroxidases and glutaredoxins, as well as reacting directly with oxidants.[79] Due to its high concentration and its central role in maintaining the cell's redox Department of State, glutathione is one of the most important cellular antioxidants.[84] In close to organisms glutathione is replaced by other thiols, such as by mycothiol in the Actinomycetes, bacillithiol in some Gram-positive bacteria,[86] [87] or past trypanothione in the Kinetoplastids.[88] [89]
E [edit out]
Vitamin E is the aggregated name for a set of eight related tocopherols and tocotrienols, which are fat-soluble vitamins with antioxidant properties.[90] [91] Of these, α-E has been most studied A IT has the highest bioavailability, with the torso preferentially absorbing and metabolising this form.[92]
It has been claimed that the α-E form is the virtually beta lipid-soluble antioxidant, and that it protects membranes from oxidisation by reacting with lipid radicals produced in the lipid peroxidation chain reaction.[90] [93] This removes the free radical intermediates and prevents the propagation reaction from continuing. This reaction produces oxidised α-tocopheroxyl radicals that commode be recycled rearmost to the active reduced form through reduction by other antioxidants, such arsenic ascorbate, retinol or ubiquinol.[94] This is in line of work with findings showing that α-tocopherol, simply non water-soluble antioxidants, expeditiously protects glutathione peroxidase 4 (GPX4)-poor cells from cell death.[95] GPx4 is the only well-known enzyme that with efficiency reduces lipoid-hydroperoxides within biologic membranes.
Yet, the roles and importance of the various forms of vitamin E are presently unclear,[96] [97] and IT has even been suggested that the most important function of α-tocopherol is as a signaling molecule, with this molecule having no significant role in antioxidant metastasis.[98] [99] The functions of the other forms of E are even less well understood, although γ-vitamin E is a nucleophile that may react with electrophilic mutagens,[92] and tocotrienols Crataegus oxycantha be important in protecting neurons from damage.[100]
In favor-oxidant activities [edit]
Antioxidants that are reducing agents fire also act pro-oxidants. E.g., vitamin C has antioxidant activity when it reduces oxidizing substances much as peroxide;[101] however, it will also reduce metal ions that generate free radicals through the Fenton reaction.[35] [102]
- 2 Fe3+ + Ascorbate → 2 Fe2+ + Dehydroascorbate
- 2 Fe2+ + 2 H2O2 → 2 Fe3+ + 2 OH· + 2 OH−
The relative importance of the antioxidant and pro-oxidant activities of antioxidants is an area of current research, but vitamin C, which exerts its personal effects as a vitamin by oxidizing polypeptides, appears to have a largely antioxidant action in the imperfect body.[102]
Enzyme systems [edit]
Enzymatic pathway for detoxification of reactive oxygen species
As with the chemical antioxidants, cells are fortified against aerobic stress by an interacting network of antioxidant enzymes.[30] [31] Here, the superoxide released by processes such as aerophilous phosphorylation is firstly converted to peroxide and then foster reduced to give water. This detoxification nerve pathway is the result of multiple enzymes, with superoxide anion dismutases catalysing the number 1 step and then catalases and various peroxidases removing peroxide. As with antioxidant metabolites, the contributions of these enzymes to antioxidant defenses can live hard to separate from one another, but the generation of transgenic mice lacking just one antioxidant enzyme bottom equal informative.[103]
SOD, catalase, and peroxiredoxins [edit]
Superoxide anion dismutases (SODs) are a class of close related enzymes that catalyze the breakdown of the superoxide anion into O and hydrogen hydrogen peroxide.[104] [105] SOD enzymes are present in well-nig all aerobic cells and in living thing fluids.[106] Superoxide dismutase enzymes contain metal ion cofactors that, conditional the isozyme, put up be copper, zinc, atomic number 25 or iron. In humans, the copper/zinc SOD is present in the cytosol, while manganese SOD is salute in the mitochondrion.[105] There also exists a third form of SOD in animate thing fluids, which contains copper and zinc in its active sites.[107] The mitochondrial isozyme seems to be the most biologically world-shattering of these three, since mice lacking this enzyme choke soon afterwards birth.[108] In contrast, the mice lacking pig/atomic number 30 SOD (Sod1) are viable but have numerous pathologies and a reduced life (see clause on superoxide anion), piece mice without the extracellular SOD rich person negligible defects (aware to hyperoxia).[103] [109] In plants, SOD isozymes are present in the cytosol and mitochondria, with an iron SOD found in chloroplasts that is absent from vertebrates and barm.[110]
Catalases are enzymes that catalyse the rebirth of peroxide to water and oxygen, victimization either an iron or manganese cofactor.[111] [112] This protein is localized to peroxisomes in most eukaryotic cells.[113] Catalase is an fantastical enzyme since, although hydrogen peroxide is its only substrate, it follows a ping-niff mechanism. Here, its cofactor is oxidised by one atom of hydrogen hydrogen peroxide and past regenerated by transferring the destined oxygen to a second molecule of substratum.[114] Despite its apparent importance in peroxide removal, humans with genetic insufficiency of catalase — "acatalasemia" — or mice genetically engineered to deficiency catalase completely, suffer some rickety effects.[115] [116]

Peroxiredoxins are peroxidases that catalyse the reduction of hydrogen peroxide, organic hydroperoxides, besides American Samoa peroxynitrite.[118] They are divided into three classes: characteristic 2-cysteine peroxiredoxins; atypical 2-cysteine peroxiredoxins; and 1-cysteine peroxiredoxins.[119] These enzymes share the same basic catalytic mechanism, in which a redox-surface-active cysteine (the peroxidatic cysteine) in the active place is oxidized to a sulfenic acid by the hydrogen peroxide substrate.[120] Over-oxidation of this cysteine residue in peroxiredoxins inactivates these enzymes, but this can be turned away the action of sulfiredoxin.[121] Peroxiredoxins seem to be great in antioxidant metabolism, as mice lacking peroxiredoxin 1 or 2 have short lifetime and suffer from hemolytic anaemia, piece plants use peroxiredoxins to transfer hydrogen peroxide generated in chloroplasts.[122] [123] [124]
Thioredoxin and glutathione systems [edit]
The thioredoxin system contains the 12-kDa protein thioredoxin and its companion thioredoxin reductase.[125] Proteins related to thioredoxin are present all told sequenced organisms. Plants, such as Arabidopsis thaliana, take over a particularly great diversity of isoforms.[126] The acrobatic site of thioredoxin consists of two abutting cysteines, as part of a highly preserved CXXC motif, that can oscillation between an active dithiol form (reduced) and an oxidised disulfide form. In its active state, thioredoxin acts as an efficient reducing agent, scavenging reactive oxygen species and maintaining other proteins in their reduced state.[127] After being oxidized, the active thioredoxin is regenerated by the action of thioredoxin reductase, victimisation NADPH as an electron donor.[128]
The glutathione organisation includes glutathione, glutathione reductase, glutathione peroxidases, and glutathione S-transferases.[84] This system is recovered in animals, plants and microorganisms.[84] [129] Glutathione peroxidase is an enzyme containing four selenium-cofactors that catalyzes the equipment failure of atomic number 1 hydrogen peroxide and constituent hydroperoxides. There are at least four different glutathione peroxidase isozymes in animals.[130] Glutathione peroxidase 1 is the almost lush and is a real efficient scavenger of hydrogen peroxide, patc glutathione peroxidase 4 is most energetic with lipid hydroperoxides. Surprisingly, glutathione peroxidase 1 is dispensable, as mice inadequate this enzyme bear modal lifespans,[131] but they are hypersensitive to induced oxidative stress.[132] In addition, the glutathione S-transferases show senior high school natural process with lipid peroxides.[133] These enzymes are at especially high-topped levels in the colorful and also serve in detoxification metabolism.[134]
Uses in technology [edit]
Intellectual nourishment preservatives [edit]
| | This segment is missing entropy about in depth sauteing. (December 2021) |
Antioxidants are used every bit food additives to help defend against food for thought deterioration. Exposure to oxygen and sunlight are the 2 main factors in the oxidation of nutrient, and then intellectual nourishment is preserved by keeping in the dark and sealing it in containers or even coating it in wax, as with cucumbers. Even so, every bit oxygen is also important for plant respiration, storing plant materials in anaerobiotic conditions produces sharp-worded flavors and unappealing colors.[135] Consequently, packaging of fresh fruits and vegetables contains an ~8% oxygen atmosphere. Antioxidants are an especially important class of preservatives as, unlike bacterial or plant spoilage, oxidation reactions unruffled occur relatively rapidly in frozen or refrigerated food.[136] These preservatives let in natural antioxidants such as ascorbic battery-acid (AA, E300) and tocopherols (E306), likewise as synthetic antioxidants such as propyl group gallate (PG, E310), tertiary butylhydroquinone (TBHQ), butylated hydroxyanisole (BHA, E320) and butylated hydroxytoluene (BHT, E321).[137] [138]
The most common molecules attacked by oxidation are unsaturated fats; oxidation causes them to turn stale.[139] Since oxidized lipids are often discolored and usually have unpleasant tastes so much as metallic or sulfurous flavors, it is valuable to avoid oxidation in fat-rich foods. So, these foods are rarely preserved by drying; instead, they are retained by smoking, salting or fermen. Even to a lesser extent butterball foods much as fruits are sprayed with sulfurous antioxidants prior to air drying. Oxidation is often catalyzed by metals, which is wherefore fats such as butter should never be intent in aluminium foil or kept in metal-looking containers. Some fatty foods such as chromatic embrocate are partially protected from oxidation by their raw content of antioxidants, but remain light-sensitive to photooxidation.[140] Antioxidant preservatives are also added to fat based cosmetics such as lipstick and moisturizers to prevent rancidity.[ citation needed ]
Industrial uses [edit]

Substituted phenols and derivatives of phenylenediamine are common antioxidants wont to inhibit gum formation in gasoline (gasolene).
Antioxidants are frequently added to business enterprise products. A common use is as stabilizers in fuels and lubricants to forestall oxidation, and in gasolines to prevent the polymerization that leads to the formation of engine-fouling residues.[141] In 2014, the planetary market for natural and synthetic antioxidants was The States$2.25 billion with a forecast of growth to $3.25 billion by 2020.[142]
Antioxidant polymer stabilizers are widely wont to prevent the degradation of polymers such as rubbers, plastics and adhesives that causes a loss of durability and flexibility in these materials.[143] Polymers containing double bonds in their main chains, such as India rubber and polybutadiene, are specially susceptible to oxidization and ozonolysis. They can be protected by antiozonants. Solid polymer products start to crack on exposed surfaces equally the material degrades and the chains break. The mode of cracking varies between atomic number 8 and ozone attack, the former causing a "crazy paving" effect, patc ozone onrush produces deeper cracks allied at opportune angles to the tensile form in the product. Oxidation and UV degradation are also often linked, mainly because UV radiation creates free radicals by in bondage breakage. The free of radicals then react with oxygen to bring out peroxy radicals which induce notwithstandin further damage, often in a chain reaction. Other polymers susceptible to oxidation admit polypropylene and polyethylene. The former is more sensitive owing to the presence of second-string carbon atoms present in every replicate unit. Attack occurs at this point because the free radical formed is more stable than one formed connected a primary carbon atom. Oxidation of polyethylene tends to occur at weak links in the chain, so much as branch points in low-density polyethylene.[ citation necessary ]
| Fuel additive | Components[144] | Applications[144] |
|---|---|---|
| AO-22 | N,N'-di-2-butyl-1,4-phenylenediamine | Turbine oils, transformer oils, hydraulic fluids, waxes, and greases |
| AO-24 | N,N'-di-2-butyl-1,4-phenylenediamine | Low-temperature oils |
| AO-29 | 2,6-di-tert-butyl-4-methylphenol | Turbine oils, transformer oils, hydraulic fluids, waxes, greases, and gasolines |
| AO-30 | 2,4-dimethyl-6-tert-butylphenol | Jet fuels and gasolines, including aviation gasolines |
| AO-31 | 2,4-dimethyl-6-tert-butylphenol | Jet fuels and gasolines, including aviation gasolines |
| AO-32 | 2,4-dimethyl-6-tert-butylphenol and 2,6-di-tert-butyl-4-methylphenol | Super C fuels and gasolines, including airmanship gasolines |
| AO-37 | 2,6-di-tert-butylphenol | Jet fuels and gasolines, widely approved for airmanship fuels |
Levels in food [edit]

Fruits and vegetables are good sources of antioxidant vitamins C and E.
Antioxidant vitamins are found in vegetables, fruits, egg, legumes and nuts. Vitamins A, C, and E can be annihilated past long-term repositing or prolonged cooking.[145] The personal effects of cooking and food for thought processing are complex, as these processes can also increase the bioavailability of antioxidants, such arsenic some carotenoids in vegetables.[146] Computerized food contains fewer antioxidant vitamins than fresh and raw foods, As preparation exposes food to heat and oxygen.[147]
| Antioxidant vitamins | Foods containing high levels of antioxidant vitamins[21] [148] [149] |
|---|---|
| Vitamin C (ascorbic acid) | Fresh operating room frozen fruits and vegetables |
| Tocopherol (tocopherols, tocotrienols) | Garlic-like oils, nuts, and seeds |
| Carotenoids (carotenes as provitamin A) | Yield, vegetables and eggs |
Other antioxidants are non obtained from the diet, only instead are made in the body. For example, ubiquinol (coenzyme Q) is poorly captive from the gut and is successful through the mevalonate pathway.[63] Some other example is glutathione, which is made from amino acids. As any glutathione in the gut is broken down to sovereign cysteine, Glycine and glutamic acid before existence attentive, still large oral examination intake has little effect on the concentration of glutathione in the body.[150] [151] Although large amounts of sulfur-containing amino acids such as acetylcysteine put up increase glutathione,[152] nary demonstrate exists that feeding high levels of these glutathione precursors is beneficial for healthy adults.[153]
Measurement and invalidation of ORAC [edit]
Measurement of polyphenol and carotenoid content in food is not a straightforward process, as antioxidants conjointly are a diverse group of compounds with different reactivities to various reactive oxygen species. In food science analyses in vitro, the oxygen radical absorbance capacity (ORAC) was once an industry standard for estimating antioxidant strength of completely foods, juices and intellectual nourishment additives, mainly from the presence of polyphenols.[154] [155] Earlier measurements and ratings by the US USDA were withdrawn in 2012 atomic number 3 biologically irrelevant to human health, referring to an petit mal epilepsy of physiological evidence for polyphenols having antioxidant properties in vivo.[156] Therefore, the ORAC method acting, derived only from in vitro experiments, is no more considered relevant to hominid diets operating room biological science, arsenic of 2010.[156]
Alternative in vitro measurements of antioxidant content in foods – also supported on the presence of polyphenols – include the Folin-Ciocalteu reagent, and the Trolox equivalent antioxidant capacity assay.[157]
Account [edit]
As persona of their adaptation from marine life, terrestrial plants began producing non-marine antioxidants such arsenic ascorbic acid (vitamin C), polyphenols and tocopherols. The evolution of angiosperm plants between 50 and 200 million years ago resulted in the development of many antioxidant pigments – specially during the Jurassic period period – as chemical defences against reactive oxygen species that are byproducts of photosynthesis.[158] Originally, the term antioxidant specifically referred to a chemical that prevented the use of goods and services of oxygen. In the late 19th and early 20th centuries, extensive study concentrated connected the use of antioxidants in immodest industrial processes, such arsenic the prevention of argentiferous corrosion, the vulcanization of rubber, and the polymerization of fuels in the fouling of internal combustion engines.[159]
Early research connected the role of antioxidants in biota focused connected their use in preventing the oxidation of unsaturated fats, which is the cause of rancidity.[160] Antioxidant body process could be premeditated simply aside placing the fat in a closed container with oxygen and measure the rate of oxygen consumption. Withal, it was the designation of vitamins C and E as antioxidants that revolutionized the field and led to the realization of the importance of antioxidants in the biochemistry of living organisms.[161] [162] The possible mechanisms of action at law of antioxidants were first explored when it was recognized that a substance with anti-oxidative activity is likely to be one that is itself readily oxidised.[163] Research into how vitamin E prevents the outgrowth of lipide peroxidation led to the identification of antioxidants As reducing agents that prevent oxidative reactions, often past scavenging reactive oxygen species before they can damage cells.[164]
References [edit]
- ^ Dabelstein W, Reglitzky A, Schütze A, Reders K (2007). "Automotive Fuels". Ullmann's Encyclopedia of Developed Alchemy. doi:10.1002/14356007.a16_719.pub2. ISBN978-3-527-30673-2.
- ^ "Antioxidants: In Profoundness". NCCIH. November 2013. Retrieved 17 April 2021.
- ^ Bjelakovic G, Nikolova D, Gluud C (2013). "Meta-regression analyses, meta-analyses, and trial sequential analyses of the effects of supplementation with beta-carotene, vitamin A, and vitamin E severally OR in different combinations on all-movement mortality: Doctor of Osteopathy we have got demonstrate for lack of harm?". PLOS ONE. 8 (9): e74558. Bibcode:2013PLoSO...874558B. doi:10.1371/diary.pone.0074558. PMC3765487. PMID 24040282.
- ^ Abner EL, Schmitt FA, Mendiondo MS, Marcum JL, Kryscio RJ (July 2011). "Vitamin E and entirely-cause fatality rate: a meta-analysis". Present-day Aging Science. 4 (2): 158–70. DoI:10.2174/1874609811104020158. PMC4030744. PMID 21235492.
- ^ Cortés-Jofré M, Rueda JR, Corsini-Muñoz G, Fonseca-Cortés C, Caraballoso M, Bonfill Cosp X (2012). "Drugs for preventing lung cancer in healthy people". The Cochrane Database of Systematic Reviews. 10: CD002141. doi:10.1002/14651858.CD002141.pub2. PMID 23076895.
- ^ Jiang L, Yang KH, Tian JH, Guan QL, Yao N, Cao N, Mi DH, Wu dialect J, Ma B, Yang SH (2010). "Efficacy of antioxidant vitamins and selenium supplement in prostate cancer prevention: a meta-analysis of randomized controlled trials". Nutrition and Cancer. 62 (6): 719–27. doi:10.1080/01635581.2010.494335. PMID 20661819. S2CID 13611123.
- ^ Rees K, Hartley L, Day C, Flowers N, Clarke A, Stranges S (2013). "Selenium supplement for the primary feather bar of cardiovascular disease" (PDF). The Cochrane Database of Systematic Reviews. 1 (1): CD009671. doi:10.1002/14651858.CD009671.pub2. PMC7433291. PMID 23440843. Archived from the original (PDF) on 12 August 2017. Retrieved 23 July 2018.
- ^ Shekelle PG, Morton Security Council, Jungvig LK, Udani J, Spar M, Tu W, J Suttorp M, Coulter I, Newberry SJ, Manly M (April 2004). "Result of secondary vitamin E for the bar and treatment of cardiovascular disease". Daybook of General Interior Medicine. 19 (4): 380–9. doi:10.1111/j.1525-1497.2004.30090.x. PMC1492195. PMID 15061748.
- ^ a b c Stanner SA, Hughes J, Kelly CN, Buttriss J (May 2004). "A review of the epidemiological evidence for the 'antioxidant hypothesis'". Common Health Nutrition. 7 (3): 407–22. doi:10.1079/PHN2003543. PMID 15153272.
- ^ a b Shenkin A (Feb 2006). "The key theatrical role of micronutrients". Clinical Nutrition. 25 (1): 1–13. Department of the Interior:10.1016/j.clnu.2005.11.006. PMID 16376462.
- ^ Woodside JV, McCall D, McGartland C, Young IS (Nov 2005). "Micronutrients: dietary intake v. supplement use". The Proceedings of the Nutrition Society. 64 (4): 543–53. doi:10.1079/PNS2005464. PMID 16313697.
- ^ Food, Nutrition, Physical Action, and the Bar of Cancer: a Global View. Cosmos Cancer Research Monetary fund (2007). ISBN 978-0-9722522-2-5.
- ^ Hail N, Cortes M, Sir Francis Drake EN, Spallholz JE (July 2008). "Cancer chemoprevention: a radical perspective". Free Basal Biology & Medication. 45 (2): 97–110. Interior Department:10.1016/j.freeradbiomed.2008.04.004. PMID 18454943.
- ^ "Flavonoids". Linus Pauling Institute, Beaver State State University, Corvallis. 2016. Retrieved 24 July 2016.
- ^ Lemmo W (September 2014). "Potential interactions of prescription and terminated-the-counter medications having antioxidant capabilities with radiation therapy and chemotherapy". International Daybook of Cancer. 137 (11): 2525–33. doi:10.1002/ijc.29208. PMID 25220632. S2CID 205951215.
- ^ Hurrell RF (September 2003). "Influence of vegetable protein sources on hint element and mineral bioavailability". The Diary of Sustenance. 133 (9): 2973S–7S. Department of the Interior:10.1093/jn/133.9.2973S. PMID 12949395.
- ^ Track dow JR (September 2003). "Bioavailability of iron, zinc, and former trace minerals from vegetarian diets". The American Journal of Clinical Nutrition. 78 (3 Suppl): 633S–639S. doi:10.1093/ajcn/78.3.633S. PMID 12936958.
- ^ Gibson RS, Perlas L, Hotz C (May 2006). "Up the bioavailability of nutrients in plant foods at the household level". The Proceeding of the Nutrition Society. 65 (2): 160–8. doi:10.1079/PNS2006489. PMID 16672077.
- ^ a b Mosha TC, Gaga HE, Pace RD, Laswai HS, Mtebe K (June 1995). "Set up of blanching on the content of antinutritional factors in hand-picked vegetables". Found Foods for Human Nutrition. 47 (4): 361–7. doi:10.1007/BF01088275. PMID 8577655. S2CID 1118651.
- ^ Sandberg AS (December 2002). "Bioavailability of minerals in legumes". The Island Journal of Nutrition. 88 Suppl 3 (Suppl 3): S281–5. doi:10.1079/BJN/2002718. PMID 12498628.
- ^ a b Beecher GR (October 2003). "Overview of dietary flavonoids: nomenclature, occurrence and intake". The Diary of Nutrition. 133 (10): 3248S–3254S. Interior Department:10.1093/jn/133.10.3248S. PMID 14519822.
- ^ Omenn GS, Goodman GE, Thornquist MD, Balmes J, Cullen MR, Glass A, Keogh JP, Meyskens FL, Valanis B, Williams JH, Barnhart S, Cherniack MG, Brodkin California, Hammar S (November 1996). "Risk factors for lung cancer and for intervention personal effects in CARET, the Beta-Carotene and Retinol Efficaciousness Trial" (PDF). Journal of the National Cancer Institute. 88 (21): 1550–9. doi:10.1093/jnci/88.21.1550. PMID 8901853.
- ^ Albanes D (June 1999). "Beta-carotene and lung Cancer: a vitrine study". The Terra firma Diary of Clinical Nutrition. 69 (6): 1345S–50S. Department of the Interior:10.1093/ajcn/69.6.1345S. PMID 10359235.
- ^ a b Bjelakovic G, Nikolova D, Gluud LL, Simonetti RG, Gluud C (February 2007). "Death rate in irregular trials of antioxidant supplements for primary and secondary prevention: systematic look back and meta-psychoanalysis". JAMA. 297 (8): 842–57. doi:10.1001/jama.297.8.842. PMID 17327526.
- ^ a b Bjelakovic G, Nikolova D, Gluud LL, Simonetti Roentgenium, Gluud C (14 March 2012). "Antioxidant supplements for prevention of mortality in healthy participants and patients with various diseases". The Cochrane Database of Tabular Reviews. 2012 (3): CD007176. doi:10.1002/14651858.CD007176.pub2. high-density lipoprotein:10138/136201. PMC8407395. PMID 22419320.
- ^ Study Citing Antioxidant Vitamin Risks Based Along Flawed Methodological analysis, Experts Argue Intelligence release from Oregon State University published on ScienceDaily. Retrieved 19 April 2007
- ^ Miller Emergency room, Pastor-Barriuso R, Dalal D, Riemersma RA, Appel LJ, Guallar E (January 2005). "Meta-analysis: high-dosage vitamin E supplementation may step-up every-campaign mortality". Annals of General medicine. 142 (1): 37–46. Interior:10.7326/0003-4819-142-1-200501040-00110. PMID 15537682.
- ^ a b Bjelakovic G, Nagorni A, Nikolova D, Simonetti RG, Bjelakovic M, Gluud C (July 2006). "Meta-analysis: antioxidant supplements for primary and secondary prevention of colorectal adenoma". Alimentary Pharmacology & Therapeutics. 24 (2): 281–91. doi:10.1111/j.1365-2036.2006.02970.x. PMID 16842454. S2CID 20452618.
- ^ Cortés-Jofré, Marcela; Rueda, José-Ramón; Asenjo-Lobos, Claudia; Madrid, Eva; Bonfill Cosp, Xavier (4 March 2020). "Drugs for preventing lung cancer in able people". The Cochrane Database of Nonrandom Reviews. 2020 (3): CD002141. Department of the Interior:10.1002/14651858.CD002141.pub3. ISSN 1469-493X. PMC7059884. PMID 32130738.
- ^ a b c Davies KJ (1995). "Oxidative accentuate: the paradox of aerobic biography". Biochemical Society Symposium. 61: 1–31. doi:10.1042/bss0610001. PMID 8660387.
- ^ a b c d e Sies H (Marchland 1997). "Oxidative stress: oxidants and antioxidants". Experimental Physiology. 82 (2): 291–5. doi:10.1113/expphysiol.1997.sp004024. PMID 9129943. S2CID 20240552.
- ^ a b c d Vertuani S, Angusti A, Manfredini S (2004). "The antioxidants and pro-antioxidants network: an overview". Underway Pharmaceutical Innovation. 10 (14): 1677–94. doi:10.2174/1381612043384655. PMID 15134565.
- ^ Rhee Atomic number 10 (June 2006). "Cellular phone signaling. H2O2, a necessary immoral for cell signaling". Scientific discipline. 312 (5782): 1882–3. doi:10.1126/scientific discipline.1130481. PMID 16809515. S2CID 83598498.
- ^ Valko M, Leibfritz D, Moncol J, Cronin Element 109, Mazur M, Telser J (2007). "Free radicals and antioxidants in normal physiological functions and human disease". The Internationalistic Journal of Biochemistry &A; Electric cell Biology. 39 (1): 44–84. doi:10.1016/j.biocel.2006.07.001. PMID 16978905.
- ^ a b Stohs SJ, Bagchi D (February 1995). "Oxidative mechanisms in the toxicity of metal ions" (PDF). Free Radical Biology & Medicine (Submitted manuscript). 18 (2): 321–36. CiteSeerX10.1.1.461.6417. doi:10.1016/0891-5849(94)00159-H. PMID 7744317.
- ^ Nakabeppu Y, Sakumi K, Sakamoto K, Tsuchimoto D, Tsuzuki T, Nakatsu Y (April 2006). "Mutagenesis and carcinogenesis caused by the oxidation of nucleic acids". Biological Chemistry. 387 (4): 373–9. doi:10.1515/BC.2006.050. PMID 16606334. S2CID 20217256.
- ^ Valko M, Izakovic M, Mazur M, Rhodes CJ, Telser J (Nov 2004). "Use of oxygen radicals in DNA scathe and cancer incidence". Molecular and Cellular Biochemistry. 266 (1–2): 37–56. doi:10.1023/B:MCBI.0000049134.69131.89. PMID 15646026. S2CID 207547763.
- ^ Stadtman ER (August 1992). "Protein oxidation and senescent". Skill. 257 (5074): 1220–4. Bibcode:1992Sci...257.1220S. doi:10.1126/science.1355616. PMID 1355616.
- ^ Raha S, Esme Stuart Lennox Robinson BH (October 2000). "Mitochondria, oxygen free radicals, disease and ageing". Trends in Biochemical Sciences. 25 (10): 502–8. doi:10.1016/S0968-0004(00)01674-1. PMID 11050436.
- ^ Lenaz G (2001). "The mitochondrial production of reactive oxygen species: mechanisms and implications in human pathology". IUBMB Spirit. 52 (3–5): 159–64. doi:10.1080/15216540152845957. PMID 11798028. S2CID 45366190.
- ^ Finkel T, Holbrook NJ (November 2000). "Oxidants, aerobic stress and the biota of ageing". Nature. 408 (6809): 239–47. Bibcode:2000Natur.408..239F. doi:10.1038/35041687. PMID 11089981. S2CID 2502238.
- ^ Hirst J, King MS, Pryde KR (October 2008). "The product of thermolabile oxygen species by complex I". Biochemical Society Minutes. 36 (Pt 5): 976–80. doi:10.1042/BST0360976. PMID 18793173.
- ^ Seaver LC, Imlay JA (November 2004). "Are respiratory enzymes the special sources of intracellular hydrogen peroxide?". The Diary of Biological Chemistry. 279 (47): 48742–50. doi:10.1074/jbc.M408754200. PMID 15361522.
- ^ a b Imlay JA (2003). "Pathways of oxidative damage". Annual Review of Microbiology. 57: 395–418. doi:10.1146/annurev.micro.57.030502.090938. PMID 14527285.
- ^ Demmig-Adams B, Adams WW (December 2002). "Antioxidants in photosynthesis and human alimentation". Science. 298 (5601): 2149–53. Bibcode:2002Sci...298.2149D. Interior Department:10.1126/skill.1078002. PMID 12481128. S2CID 27486669.
- ^ Krieger-Liszkay A (January 2005). "Vest oxygen production in photosynthesis". Journal of Experimental Botany. 56 (411): 337–46. CiteSeerX10.1.1.327.9651. doi:10.1093/jxb/erh237. PMID 15310815.
- ^ Kupper FC, Carpenter LJ, McFiggans GB, Palmer CJ, Waite TJ, Boneberg E-M, Woitsch S, Weiller M, Abela R, Grolimund D, Potin P, Butler A, Luther GW, Kroneck PMH, Meyer-Klaucke W, Feiters MC (2008). "Iodide accruement provides kelp with an inorganic antioxidant impacting atmospheric chemistry". Proceedings of the National Academy of Sciences. 105 (19): 6954–6958. Bibcode:2008PNAS..105.6954K. doi:10.1073/pnas.0709959105. ISSN 0027-8424. PMC2383960. PMID 18458346.
- ^ Szabó I, Bergantino E, Giacometti GM (July 2005). "Light and oxygenic photosynthesis: energy dissipation as a protection mechanism against photo-oxidation". EMBO Reports. 6 (7): 629–34. doi:10.1038/sj.embor.7400460. PMC1369118. PMID 15995679.
- ^ Kerfeld CA (October 2004). "Soluble carotenoid proteins of cyanobacteria" (PDF). Archives of Biochemistry and Biophysics (Submitted manuscript). 430 (1): 2–9. doi:10.1016/j.abb.2004.03.018. PMID 15325905.
- ^ Miller RA, Britigan BE (January 1997). "Role of oxidants in microbial pathophysiology". Medical institution Microbiology Reviews. 10 (1): 1–18. doi:10.1128/CMR.10.1.1. PMC172912. PMID 8993856.
- ^ Chaudière J, Ferrari-Iliou R (1999). "Animate thing antioxidants: from chemical to biochemical mechanisms". Solid food and Chemical Toxicology. 37 (9–10): 949–62. doi:10.1016/S0278-6915(99)00090-3. PMID 10541450.
- ^ Sies H (July 1993). "Strategies of antioxidant demurrer". European Journal of Biochemistry. 215 (2): 213–9. Interior:10.1111/j.1432-1033.1993.tb18025.x. PMID 7688300.
- ^ Khaw KT, Woodhouse P (June 1995). "Interrelation of ascorbic acid, infection, haemostatic factors, and cardiovascular disease". BMJ. 310 (6994): 1559–63. doi:10.1136/bmj.310.6994.1559. PMC2549940. PMID 7787643.
- ^ a b c d Evelson P, Travacio M, Repetto M, Escobar J, Llesuy S, Lissi Ea (April 2001). "Evaluation of total reactive antioxidant potential (TRAP) of tissue homogenates and their cytosols". Archives of Biochemistry and Biophysics. 388 (2): 261–6. doi:10.1006/abbi.2001.2292. PMID 11368163.
- ^ Morrison JA, Jacobsen DW, Sprecher DL, Robinson K, Khoury P, Daniels Strontium (Nov 1999). "Serum glutathione in adolescent males predicts parental anatomical structure heart disease". Circulation. 100 (22): 2244–7. doi:10.1161/01.CIR.100.22.2244. PMID 10577998.
- ^ Teichert J, Preiss R (November 1992). "HPLC-methods for determination of lipoic acid and its faded form in human plasma". International Journal of Clinical Pharmacological medicine, Therapy, and Toxicology. 30 (11): 511–2. PMID 1490813.
- ^ Akiba S, Matsugo S, Packer L, Konishi T (May 1998). "Assay of protein-bound lipoic acid in tissues by a sunrise catalyst method". Analytical Biochemistry. 258 (2): 299–304. doi:10.1006/abio.1998.2615. PMID 9570844.
- ^ a b Glantzounis GK, Tsimoyiannis EC, Kappas AM, Galaris District attorney (2005). "Uric acid and oxidative emphasis". Current Medicament Design. 11 (32): 4145–51. doi:10.2174/138161205774913255. PMID 16375736.
- ^ Elevated railroad-Sohemy A, Baylin A, Kabagambe E, Ascherio A, Spiegelman D, Campos H (July 2002). "Individual carotenoid concentrations in fatty tissue and plasma as biomarkers of dietary intake". The American Daybook of Clinical Alimentation. 76 (1): 172–9. doi:10.1093/ajcn/76.1.172. PMID 12081831.
- ^ a b Sowell Aluminium, Huff DL, Yeager PR, Caudill SP, Gunter EW (March 1994). "Retinol, alpha-tocopherol, lutein/zeaxanthin, beta-cryptoxanthin, lycopene, alpha-carotene, trans-beta-carotene, and quaternion retinyl esters in blood serum determined at the same time by reversed-form HPLC with multiwavelength espial". Clinical Chemistry. 40 (3): 411–6. DoI:10.1093/clinchem/40.3.411. PMID 8131277.
- ^ Stahl W, Schwarz W, Sundquist AR, Sies H (April 1992). "cis-trans isomers of lycopene and beta-carotene in human blood serum and tissues". Archives of Biochemistry and Biophysics. 294 (1): 173–7. doi:10.1016/0003-9861(92)90153-N. PMID 1550343.
- ^ Zita C, Overvad K, Mortensen SA, Sindberg CD, Moesgaard S, Hunter DA (2003). "Serum coenzyme Q10 concentrations in healthy men supplemented with 30 mg Oregon 100 mg coenzyme Q10 for deuce months in a randomised controlled study". BioFactors. 18 (1–4): 185–93. doi:10.1002/biof.5520180221. PMID 14695934. S2CID 19895215.
- ^ a b Turunen M, Olsson J, Dallner G (Jan 2004). "Metabolism and function of coenzyme Q". Biochimica et Biophysica Acta (BBA) - Biomembranes. 1660 (1–2): 171–99. doi:10.1016/j.bbamem.2003.11.012. PMID 14757233.
- ^ a b Enomoto A, Endou H (September 2005). "Roles of organic anion transporters (OATs) and a urate transporter (URAT1) in the pathophysiology of human disease". Clinical and Experimental Nephrology. 9 (3): 195–205. Interior Department:10.1007/s10157-005-0368-5. PMID 16189627. S2CID 6145651.
- ^ a b Wu XW, Leeward CC, Muzny DM, Caskey CT (December 1989). "Urate oxidase: capital structure and evolutionary implications". Proceedings of the National Academy of Sciences of the U.S.A. 86 (23): 9412–6. Bibcode:1989PNAS...86.9412W. Interior:10.1073/pnas.86.23.9412. PMC298506. PMID 2594778.
- ^ Wu XW, Muzny Diabetes mellitus, Lee CC, Caskey CT (January 1992). "Two independent mutational events in the red ink of urate oxidase during hominoid phylogeny". Journal of Unit Development. 34 (1): 78–84. Bibcode:1992JMolE..34...78W. doi:10.1007/BF00163854. PMID 1556746. S2CID 33424555.
- ^ Álvarez-Lario B, Macarrón-Vicente J (November 2010). "Excreta acid and evolution". Rheumatology. 49 (11): 2010–5. doi:10.1093/rheumatology/keq204. PMID 20627967.
- ^ a b Watanabe S, Kang DH, Feng L, Nakagawa T, Kanellis J, LAN H, Mazzali M, Johnson RJ (September 2002). "Uric acid, hominoid evolution, and the pathogenesis of salt-sensitivity". Hypertension. 40 (3): 355–60. DoI:10.1161/01.HYP.0000028589.66335.AA. PMID 12215479.
- ^ Dr. Johnson RJ, Andrews P, Benner SA, King Olive W (2010). "Theodore E. Woodward award. The phylogenesis of obesity: insights from the mid-Miocene epoch". Minutes of the American Clinical and Climatological Association. 121: 295–305, discussion 305–8. PMC2917125. PMID 20697570.
- ^ a b Baillie JK, Bates MG, Thompson AA, Waring WS, Partridge RW, Schnopp MF, Simpson A, Gulliver-Sloan F, Maxwell Strontium, Webb DJ (Crataegus oxycantha 2007). "Angiosperm urate production augments plasma antioxidant electrical capacity in healthy lowland subjects uncovered to nasal altitude". Bureau. 131 (5): 1473–8. doi:10.1378/chest.06-2235. PMID 17494796.
- ^ Becker BF (June 1993). "Towards the physiological function of uric acidic". Radical Biology &adenosine monophosphate; Medicine. 14 (6): 615–31. doi:10.1016/0891-5849(93)90143-I. PMID 8325534.
- ^ a b Sautin YY, Johnson RJ (June 2008). "Uric acid: the oxidant-antioxidant paradox". Nucleosides, Nucleotides & Nucleic Acids. 27 (6): 608–19. doi:10.1080/15257770802138558. PMC2895915. PMID 18600514.
- ^ Eggebeen AT (September 2007). "Gout: an update". American Family Physician. 76 (6): 801–8. PMID 17910294.
- ^ Campion EW, Glynn RJ, DeLabry LO (March 1987). "Asymptomatic hyperuricemia. Risks and consequences in the Normative Aging Study". The Land Journal of Music. 82 (3): 421–6. Department of the Interior:10.1016/0002-9343(87)90441-4. PMID 3826098.
- ^ Nazarewicz RR, Ziolkowski W, Vaccaro PS, Ghafourifar P (December 2007). "Event of dead-condition ketogenic diet on oxidoreduction status of human blood". Rejuvenation Explore. 10 (4): 435–40. doi:10.1089/rej.2007.0540. PMID 17663642.
- ^ a b c d "Vitamin C". Micronutrient Information Center, Linus Pauling Institute, Oregon State University, Corvallis, OR. 1 July 2018. Retrieved 19 June 2019.
- ^ Smirnoff N (2001). "L-ascorbic acid biosynthesis". Cofactor Biogenesis. Vitamins & Hormones. 61. pp. 241–66. doi:10.1016/S0083-6729(01)61008-2. ISBN978-0-12-709861-6. PMID 11153268.
- ^ Linster CL, New wave Schaftingen E (January 2007). "Vitamin C. Biosynthesis, recycling and abasement in mammals". The FEBS Journal. 274 (1): 1–22. doi:10.1111/j.1742-4658.2006.05607.x. PMID 17222174. S2CID 21345196.
- ^ a b Meister A (April 1994). "Glutathione-vitamin C antioxidant organisation in animals". The Journal of Biological Chemistry. 269 (13): 9397–400. Interior:10.1016/S0021-9258(17)36891-6. PMID 8144521.
- ^ Wells WW, Xu Displaced person, Yang YF, Rocque PA (September 1990). "Class thioltransferase (glutaredoxin) and protein disulfide isomerase have dehydroascorbate reductase activity". The Journal of Life Chemistry. 265 (26): 15361–4. doi:10.1016/S0021-9258(18)55401-6. PMID 2394726.
- ^ Padayatty SJ, Katz A, Wang Y, Eck P, Kwon O, Lee JH, Chen S, Corpe C, Dutta A, Dutta SK, Levine M (February 2003). "Ascorbic acid as an antioxidant: evaluation of its role in disease bar". Journal of the Land College of Nutrition. 22 (1): 18–35. DoI:10.1080/07315724.2003.10719272. PMID 12569111. S2CID 21196776.
- ^ Shigeoka S, Ishikawa T, Tamoi M, Miyagawa Y, Takeda T, Yabuta Y, Yoshimura K (May 2002). "Regulation and work of ascorbate peroxidase isoenzymes". Journal of Research Botany. 53 (372): 1305–19. doi:10.1093/jexbot/53.372.1305. PMID 11997377.
- ^ Smirnoff N, Wheeler GL (2000). "Ascorbic acid in plants: biosynthesis and function". Critical Reviews in Biochemistry and Unit Biology. 35 (4): 291–314. doi:10.1080/10409230008984166. PMID 11005203. S2CID 85060539.
- ^ a b c d Meister A, Anderson Pine Tree State (1983). "Glutathione". Yearly Review of Biochemistry. 52: 711–60. doi:10.1146/annurev.bi.52.070183.003431. PMID 6137189.
- ^ Meister A (November 1988). "Glutathione metabolism and its discriminating modification". The Diary of Biological Chemistry. 263 (33): 17205–8. doi:10.1016/S0021-9258(19)77815-6. PMID 3053703.
- ^ Gaballa A, Newton GL, Antelmann H, Parsonage D, Upton H, Rawat M, Claiborne A, Fahey RC, Helmann JD (April 2010). "Biogenesis and functions of bacillithiol, a major low-molecular-weight unit thiol in Bacilli". Proceeding of the National Academy of Sciences of the United States of America. 107 (14): 6482–6. Bibcode:2010PNAS..107.6482G. doi:10.1073/pnas.1000928107. PMC2851989. PMID 20308541.
- ^ Newton GL, Rawat M, La Clair JJ, Jothivasan VK, Budiarto T, Lady Emma Hamilton CJ, Claiborne A, Helmann JD, Fahey RC (Sep 2009). "Bacillithiol is an antioxidant thiol produced in Bacilli". Nature Chemical Biology. 5 (9): 625–627. doi:10.1038/nchembio.189. PMC3510479. PMID 19578333.
- ^ Fahey RC (2001). "Original thiols of prokaryotes". Period of time Review of Microbiology. 55: 333–56. doi:10.1146/annurev.micro.55.1.333. PMID 11544359.
- ^ Fairlamb AH, Cerami A (1992). "Metabolism and functions of trypanothione in the Kinetoplastida". Annual Review of Microbiology. 46: 695–729. Department of the Interior:10.1146/annurev.mi.46.100192.003403. PMID 1444271.
- ^ a b Herrera E, Barbas C (March 2001). "Tocopherol: action, metabolism and perspectives". Daybook of Physiology and Biochemistry. 57 (2): 43–56. doi:10.1007/BF03179812. hdl:10637/720. PMID 11579997. S2CID 7272312.
- ^ Packer L, Weber SU, Rimbach G (February 2001). "Molecular aspects of alpha-tocotrienol antioxidant litigate and cell signalling". The Daybook of Nutrition. 131 (2): 369S–73S. doi:10.1093/jn/131.2.369S. PMID 11160563.
- ^ a b Brigelius-Flohé R, Traber Atomic number 12 (July 1999). "Vitamin E: function and metabolism". FASEB Journal. 13 (10): 1145–55. CiteSeerX10.1.1.337.5276. doi:10.1096/fasebj.13.10.1145. PMID 10385606. S2CID 7031925.
- ^ Traber MG, Atkinson J (July 2007). "Vitamin E, antioxidant and nothing more". Free Radical Biology & Medicament. 43 (1): 4–15. doi:10.1016/j.freeradbiomed.2007.03.024. PMC2040110. PMID 17561088.
- ^ Wang X, Quinn PJ (July 1999). "Tocopherol and its function in membranes". Progress in Lipid Research. 38 (4): 309–36. doi:10.1016/S0163-7827(99)00008-9. PMID 10793887.
- ^ Seiler A, Schneider M, Förster H, Roth S, Wirth EK, Culmsee C, Plesnila N, Kremmer E, Rådmark O, Wurst W, Bornkamm GW, Schweizer U, Conrad M (Sep 2008). "Glutathione peroxidase 4 senses and translates oxidative tenseness into 12/15-lipoxygenase hanging- and AIF-mediated cell death". Cellular phone Metamorphosis. 8 (3): 237–48. doi:10.1016/j.cmet.2008.07.005. PMID 18762024.
- ^ Brigelius-Flohé R, Davies KJ (July 2007). "Is vitamin E an antioxidant, a governor of signal transduction and cistron expression, or a 'debris' food? Comments happening the cardinal accompanying papers: "Molecular mechanism of alpha-tocopherol activity" by A. Azzi and "Vitamin E, antioxidant and nothing more" by M. Traber and J. Atkinson". Free New Biology & Medicine. 43 (1): 2–3. doi:10.1016/j.freeradbiomed.2007.05.016. PMID 17561087.
- ^ Atkinson J, Epand RF, Epand RM (March 2008). "Tocopherols and tocotrienols in membranes: a critical review". Free Radical Biota & Medical specialty. 44 (5): 739–64. Department of the Interior:10.1016/j.freeradbiomed.2007.11.010. PMID 18160049.
- ^ Azzi A (July 2007). "Molecular chemical mechanism of alpha-vitamin E action". Free Radical Biology & Medicate. 43 (1): 16–21. doi:10.1016/j.freeradbiomed.2007.03.013. PMID 17561089.
- ^ Zingg JM, Azzi A (May 2004). "Non-antioxidant activities of vitamin E". Current Medicative Chemistry. 11 (9): 1113–33. doi:10.2174/0929867043365332. PMID 15134510. Archived from the originative on 6 October 2011.
- ^ Sen CK, Khanna S, Roy S (March 2006). "Tocotrienols: Vitamin E beyond tocopherols". Life Sciences. 78 (18): 2088–98. doi:10.1016/j.lfs.2005.12.001. PMC1790869. PMID 16458936.
- ^ Duarte TL, Lunec J (July 2005). "Review: When is an antioxidant non an antioxidant? A review of novel actions and reactions of vitamin C". Free Radical Explore. 39 (7): 671–86. doi:10.1080/10715760500104025. PMID 16036346. S2CID 39962659.
- ^ a b Carr A, Frei B (June 1999). "Does vitamin C act A a pro-oxidant under physiological conditions?". FASEB Journal. 13 (9): 1007–24. doi:10.1096/fasebj.13.9.1007. PMID 10336883. S2CID 15426564.
- ^ a b Ho YS, Magnenat JL, Gargano M, Cao J (October 1998). "The nature of antioxidant defense mechanisms: a deterrent example from transgenic studies". Environmental Health Perspectives. 106 Suppl 5 (Suppl 5): 1219–28. doi:10.2307/3433989. JSTOR 3433989. PMC1533365. PMID 9788901.
- ^ Zelko IN, Mariani TJ, Folz RJ (August 2002). "Superoxide dismutase multigene family: a comparison of the CuZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) gene structures, evolution, and expression". Available Form Biology &adenosine monophosphate; Medicament. 33 (3): 337–49. doi:10.1016/S0891-5849(02)00905-X. PMID 12126755.
- ^ a b Banister JV, Bannister WH, Rotilio G (1987). "Aspects of the structure, function, and applications of SOD". CRC Sarcastic Reviews in Biochemistry. 22 (2): 111–80. doi:10.3109/10409238709083738. PMID 3315461.
- ^ Johnson F, Giulivi C (2005). "Superoxide dismutases and their impact upon human health". Molecular Aspects of Medicine. 26 (4–5): 340–52. doi:10.1016/j.mam.2005.07.006. PMID 16099495.
- ^ Nozik-Grayck E, Suliman HB, Piantadosi Atomic number 20 (December 2005). "Animate thing superoxide dismutase". The International Diary of Biochemistry & Cell Biological science. 37 (12): 2466–71. doi:10.1016/j.biocel.2005.06.012. PMID 16087389.
- ^ Melov S, Schneider JA, Day BJ, Hinerfeld D, Coskun P, Mirra SS, Crapo JD, Wallace DC (February 1998). "A novel neurological phenotype in mice lacking mitochondrial manganese superoxide dismutase". Nature Genetics. 18 (2): 159–63. doi:10.1038/ng0298-159. PMID 9462746. S2CID 20843002.
- ^ Reaume AG, Elliott JL, Hoffman EK, Kowall NW, Ferrante RJ, Siwek DF, Wilcox HM, Flood DG, Beal MF, Brown RH, Scott RW, Snider WD (Whitethorn 1996). "Motor neurons in Cu/Zn superoxide dismutase-deficient mice develop unremarkably but exhibit increased necrobiosis after axonal injury". Nature Genetics. 13 (1): 43–7. Interior:10.1038/ng0596-43. PMID 8673102. S2CID 13070253.
- ^ Avant-garde Camp W, Inzé D, Van Montagu M (1997). "The regulation and function of tobacco superoxide anion dismutases". Free Radical Biology & Medicinal drug. 23 (3): 515–20. doi:10.1016/S0891-5849(97)00112-3. PMID 9214590.
- ^ Chelikani P, Fita I, Loewen PC (January 2004). "Diversity of structures and properties among catalases" (PDF). Cellular and Unit Life Sciences (Submitted manuscript). 61 (2): 192–208. doi:10.1007/s00018-003-3206-5. hdl:10261/111097. PMID 14745498. S2CID 4411482.
- ^ Zámocký M, Koller F (1999). "Understanding the structure and operate of catalases: clues from molecular evolution and in vitro mutagenesis". Progress in Biophysics and Molecular Biology. 72 (1): 19–66. doi:10.1016/S0079-6107(98)00058-3. PMID 10446501.
- ^ del Río LA, Sandalio LM, Palma JM, Bueno P, Corpas FJ (November 1992). "Metabolism of O radicals in peroxisomes and cellular implications". Free Radical Biology & Medicine. 13 (5): 557–80. DoI:10.1016/0891-5849(92)90150-F. PMID 1334030.
- ^ Hiner AN, Raven EL, Thorneley RN, García-Cánovas F, Rodríguez-López JN (July 2002). "Mechanisms of pinnated I formation in heme peroxidases". Journal of Inorganic Biochemistry. 91 (1): 27–34. doi:10.1016/S0162-0134(02)00390-2. PMID 12121759.
- ^ Mueller S, Riedel HD, Stremmel W (December 1997). "Direct evidence for catalase as the paramount H2O2 -removing enzyme in human erythrocytes". Line of descent. 90 (12): 4973–8. doi:10.1182/blood.V90.12.4973. PMID 9389716.
- ^ Ogata M (February 1991). "Acatalasemia". Human Genetics. 86 (4): 331–40. Interior:10.1007/BF00201829. PMID 1999334.
- ^ Rectory D, Youngblood D, Sarma G, Wood Z, Karplus P, Poole L (2005). "Analysis of the tie between enzymatic activity and oligomeric state in AhpC, a bacterial peroxiredoxin". Biochemistry. 44 (31): 10583–92. doi:10.1021/bi050448i. PMC3832347. PMID 16060667. PDB 1YEX
- ^ Rhee SG, Chae HZ, Kim K (June 2005). "Peroxiredoxins: a historical overview and speculative preview of novel mechanisms and emergent concepts in cadre signaling". Free Them Biology & Medicine. 38 (12): 1543–52. doi:10.1016/j.freeradbiomed.2005.02.026. PMID 15917183.
- ^ Wood ZA, Schröder E, Robin Harris J, Poole Pound (January 2003). "Structure, mechanism and regulation of peroxiredoxins". Trends in Organic chemistry Sciences. 28 (1): 32–40. DoI:10.1016/S0968-0004(02)00003-8. PMID 12517450.
- ^ Claiborne A, Yeh Malaysian Mujahidin Group, Mallett Tc, Luba J, Crane EJ, Charrier V, Parsonage D (November 1999). "Protein-sulfenic acids: diverse roles for an unlikely player in enzyme catalysis and redox regulation". Biochemistry. 38 (47): 15407–16. doi:10.1021/bi992025k. PMID 10569923.
- ^ Jönsson TJ, Lowther WT (2007). "The peroxiredoxin repair proteins". Peroxiredoxin Systems. Subcellular Biochemistry. 44. pp. 115–41. doi:10.1007/978-1-4020-6051-9_6. ISBN978-1-4020-6050-2. PMC2391273. PMID 18084892.
- ^ Neumann CA, Krause Element 110, Carman CV, Das S, Dubey DP, Abraham JL, Bronson RT, Fujiwara Y, Orkin SH, Van Etten RA (July 2003). "Essential persona for the peroxiredoxin Prdx1 in erythrocyte antioxidant defence and tumour suppression" (PDF). Nature. 424 (6948): 561–5. Bibcode:2003Natur.424..561N. doi:10.1038/nature01819. PMID 12891360. S2CID 3570549.
- ^ Lee TH, Kim SU, Yu SL, Kim SH, Park DS, Moon HB, Dho SH, Kwon KS, Kwon HJ, Han YH, Jeong S, Kang SW, Shin bone HS, Lee KK, Rhee SG, Yu DY (June 2003). "Peroxiredoxin II is essential for sustaining life span of erythrocytes in mice". Blood. 101 (12): 5033–8. doi:10.1182/descent-2002-08-2548. PMID 12586629.
- ^ Dietz KJ, Jacob S, Oelze Millilitre, Laxa M, Tognetti V, de Miranda SM, Baier M, Finkemeier I (2006). "The function of peroxiredoxins in implant cell organ oxidoreduction metabolism". Journal of Empiric Botany. 57 (8): 1697–709. Interior:10.1093/jxb/erj160. PMID 16606633.
- ^ Nordberg J, Arnér ES (December 2001). "Reactive oxygen species, antioxidants, and the mammalian thioredoxin system". Free Radical Biology & Medicine. 31 (11): 1287–312. doi:10.1016/S0891-5849(01)00724-9. PMID 11728801.
- ^ Vieira Dos Santos C, Rey P (July 2006). "Plant thioredoxins are key actors in the oxidative stress response". Trends in Plant life Skill. 11 (7): 329–34. doi:10.1016/j.tplants.2006.05.005. PMID 16782394.
- ^ Arnér ES, Holmgren A (October 2000). "Physical functions of thioredoxin and thioredoxin reductase". European Journal of Biochemistry. 267 (20): 6102–9. doi:10.1046/j.1432-1327.2000.01701.x. PMID 11012661.
- ^ Mustacich D, Powis G (February 2000). "Thioredoxin reductase". The Biochemical Journal. 346 (1): 1–8. doi:10.1042/0264-6021:3460001. PMC1220815. PMID 10657232.
- ^ Creissen G, Broadbent P, Wallace Stevens R, Wellburn AR, Mullineaux P (May 1996). "Manipulation of glutathione metabolism in transgenic plants". Biochemical Society Transactions. 24 (2): 465–9. doi:10.1042/bst0240465. PMID 8736785.
- ^ Brigelius-Flohé R (November 1999). "Weave-limited functions of individual glutathione peroxidases". Free Radical Biological science & Medicate. 27 (9–10): 951–65. doi:10.1016/S0891-5849(99)00173-2. PMID 10569628.
- ^ Ho YS, Magnenat JL, Bronson RT, Cao J, Gargano M, Sugawara M, Funk CD (June 1997). "Mice insufficient in cellular glutathione peroxidase develop commonly and show no inflated sensitivity to hyperoxia". The Journal of Biological Chemical science. 272 (26): 16644–51. Interior:10.1074/jbc.272.26.16644. PMID 9195979.
- ^ First State Haan JB, Bladier C, Griffiths P, Kelner M, O'Shea RD, Cheung NS, Bronson RT, Silvestro MJ, Angry S, Zheng SS, Beart PM, Hertzog PJ, Cola acuminat I (August 1998). "Mice with a homozygous zip genetic mutation for the near riotous glutathione peroxidase, Gpx1, show increased susceptibility to the oxidative stress-inducing agents paraquat and atomic number 1 peroxide". The Journal of Biological Chemistry. 273 (35): 22528–36. doi:10.1074/jbc.273.35.22528. PMID 9712879.
- ^ Sharma R, Yang Y, Sharma A, Awasthi S, Awasthi YC (Apr 2004). "Antioxidant role of glutathione S-transferases: protection against oxidant toxicity and regulation of stress-mediated programmed cell death". Antioxidants & Redox Signaling. 6 (2): 289–300. doi:10.1089/152308604322899350. PMID 15025930.
- ^ Hayes JD, Flanagan JU, Jowsey IR (2005). "Glutathione transferases". Period Review of Pharmacology and Toxicology. 45: 51–88. doi:10.1146/annurev.pharmtox.45.120403.095857. PMID 15822171.
- ^ Kader AA, Zagory D, Kerbel EL (1989). "Modified atmosphere promotion of fruits and vegetables". Critical Reviews in Food Science and Nutrition. 28 (1): 1–30. Interior Department:10.1080/10408398909527490. PMID 2647417.
- ^ Zallen Pica em, Hitchcock MJ, Goertz GE (December 1975). "Chilled food systems. Effects of chilled holding on quality of beef loaves". Daybook of the Dry land Dietetical Association. 67 (6): 552–7. PMID 1184900.
- ^ Iverson F (June 1995). "Phenoplast antioxidants: Wellness Protection Branch studies on butylated hydroxyanisole". Cancer Letters. 93 (1): 49–54. doi:10.1016/0304-3835(95)03787-W. PMID 7600543.
- ^ "E number exponent". UK food guide. Archived from the original on 4 March 2007. Retrieved 5 March 2007.
- ^ Robards K, Kerr AF, Patsalides E (February 1988). "Rancidity and its mensuration in edible oils and snack foods. A survey". The Psychoanalyst. 113 (2): 213–24. Bibcode:1988Ana...113..213R. Interior Department:10.1039/an9881300213. PMID 3288002.
- ^ Del Carlo M, Sacchetti G, Di Mattia C, Compagnone D, Mastrocola D, Liberatore L, Cichelli A (June 2004). "Donation of the phenolic divide to the antioxidant activeness and aerophilous stability of chromatic oil". Journal of Agricultural and Nutrient Chemistry. 52 (13): 4072–9. doi:10.1021/jf049806z. PMID 15212450.
- ^ Dipsomaniac Atomic number 58, Hammond GS, Hamilton CE, Sen JN (1955). "Air Oxidation of Hydrocarbons.1II. The Stoichiometry and Fate of Inhibitors in Benzene and Chlorobenzene". Journal of the American Chemical Society. 77 (12): 3233–7. DoI:10.1021/ja01617a026.
- ^ "Globular Antioxidants (Natural and Synthetic) Commercialise Poised to Surge From USD 2.25 Billion in 2014 to USD 3.25 Billion by 2020, Growing at 5.5% CAGR". GlobalNewswire, El Segundo, CA. 19 January 2016. Retrieved 30 January 2017.
- ^ "Why use Antioxidants?". SpecialChem Adhesives. Archived from the master copy on 11 February 2007. Retrieved 27 February 2007.
- ^ a b "Fire antioxidants". Innospec Chemicals. Archived from the original on 15 October 2006. Retrieved 27 February 2007.
- ^ Rodriguez-Amaya DB (2003). "Solid food carotenoids: analysis, composition and alterations during storage and processing of foods". Forum of Nutrition. 56: 35–7. PMID 15806788.
- ^ Maiani G, Castón MJ, Catasta G, Toti E, Cambrodón Immune gamma globulin, Bysted A, Granado-Lorencio F, Olmedilla-Alonso B, Knuthsen P, Valoti M, Böhm V, Mayer-Miebach E, Behsnilian D, Schlemmer U (September 2009). "Carotenoids: actual knowledge on food sources, intakes, stability and bioavailability and their tutelary role in humans". Molecular Nutrition &A; Food Inquiry. 53 Suppl 2: S194–218. DoI:10.1002/mnfr.200800053. hdl:10261/77697. PMID 19035552. Archived from the original on 27 September 2018. Retrieved 18 April 2017.
- ^ Henry CJ, Heppell N (February 2002). "Nutritional losses and gains during processing: rising problems and issues". The Proceedings of the Nourishment Beau monde. 61 (1): 145–8. Department of the Interior:10.1079/PNS2001142. PMID 12002789.
- ^ "Antioxidants and Cancer Bar: Fact Sheet". National Genus Cancer Constitute. Archived from the original on 4 March 2007. Retrieved 27 February 2007.
- ^ Ortega R (December 2006). "Importance of functional foods in the Mediterranean diet". Public Wellness Nutrition. 9 (8A): 1136–40. Department of the Interior:10.1017/S1368980007668530. PMID 17378953.
- ^ Witschi A, Reddy S, Stofer B, Lauterburg BH (1992). "The systemic accessibility of oral glutathione". European Diary of Objective Pharmacology. 43 (6): 667–9. doi:10.1007/BF02284971. PMID 1362956. S2CID 27606314.
- ^ Flagg EW, Coates RJ, Eley JW, Jones DP, Gunter Electronic warfare, Byers TE, Block GS, Greenberg RS (1994). "Dietary glutathione uptake in humans and the relationship 'tween intake and plasm total glutathione level". Nutrition and Crab. 21 (1): 33–46. DoI:10.1080/01635589409514302. PMID 8183721.
- ^ Dodd S, Dean O, Copolov DL, Malhi GS, Berk M (December 2008). "N-acetylcysteine for antioxidant therapy: pharmacology and clinical utility". Expert Legal opinion along Biologic Therapy. 8 (12): 1955–62. doi:10.1517/14728220802517901. PMID 18990082. S2CID 74736842.
- ^ van de Poll Megacycle per second, Dejong CH, Soeters PB (June 2006). "Fair to middling range for sulfur-containing amino acids and biomarkers for their supererogatory: lessons from enteral and parenteral nutrition". The Journal of Nutrition. 136 (6 Suppl): 1694S–1700S. doi:10.1093/jn/136.6.1694S. PMID 16702341.
- ^ Cao G, Alessio Hectometer, Cutler RG (March on 1993). "Oxygen-radical absorbance content assay for antioxidants". Free Root word Biological science & Medicine. 14 (3): 303–11. Interior:10.1016/0891-5849(93)90027-R. PMID 8458588.
- ^ Ou B, Hampsch-Woodill M, Prior RL (October 2001). "Development and validation of an built oxygen radical absorbance capacity assay using fluorescein as the colourful probe". Journal of Agricultural and Food Chemistry. 49 (10): 4619–26. doi:10.1021/jf010586o. PMID 11599998.
- ^ a b "Withdrawn: Oxygen Stem Absorbance Capacity (ORAC) of Selected Foods, Release 2 (2010)". U.S. government Section of Agriculture, Agricultural Search Service. 16 May 2012. Retrieved 13 June 2012.
- ^ Prior RL, Wu X, Schaich K (May 2005). "Replaceable methods for the determination of antioxidant capacity and phenolics in foods and dietetical supplements" (PDF). Journal of Agricultural and Intellectual nourishment Alchemy. 53 (10): 4290–302. doi:10.1021/jf0502698. PMID 15884874. Archived from the original (PDF) on 29 December 2016. Retrieved 24 October 2017.
- ^ Benzie IF (September 2003). "Phylogenesis of dietary antioxidants". Comparative Biochemistry and Physiology A. 136 (1): 113–26. Department of the Interior:10.1016/S1095-6433(02)00368-9. alpha-lipoprotein:10397/34754. PMID 14527634.
- ^ Mattill HA (1947). "Antioxidants". Annual Review of Biochemistry. 16: 177–92. doi:10.1146/annurev.bi.16.070147.001141. PMID 20259061.
- ^ German JB (1999). "Food Processing and Lipoid Oxidation". Wallop of Processing on Food for thought Safe. Advances in Experimental Medicine and Biology. 459. pp. 23–50. doi:10.1007/978-1-4615-4853-9_3. ISBN978-0-306-46051-7. PMID 10335367.
- ^ Jacob RA (1996). Three eras of vitamin C discovery. Subcellular Biochemistry. 25. pp. 1–16. Interior Department:10.1007/978-1-4613-0325-1_1. ISBN978-1-4613-7998-0. PMID 8821966.
- ^ Knight JA (1998). "Free radicals: their history and current condition in aging and disease". Annals of Clinical and Laboratory Science. 28 (6): 331–46. PMID 9846200.
- ^ Moureu C, Dufraisse C (1922). "Sur l'autoxydation: Les antioxygènes". Comptes Rendus stilboestrol Séances et Mémoires Delaware la Société de Biologie (in French). 86: 321–322.
- ^ Wolf G (March 2005). "The discovery of the antioxidant function of vitamin E: the contribution of Henry A. Mattill". The Daybook of Nourishment. 135 (3): 363–6. doi:10.1093/jn/135.3.363. PMID 15735064.
Further reading [edit]
- Halliwell, Barry. and Gospel According to John M. C. Gutteridge, Free Radicals in Biology and Medicine (Oxford University University Press, 2007), ISBN 0-19-856869-X
- Lane, Nick, Oxygen: The Molecule That Successful the Humans (Oxford University Press, 2003), ISBN 0-19-860783-0
- Pokorny, Jan, Nelly Yanishlieva, and Michael H. Gordon, Antioxidants in Food: Practical Applications (CRC Public press, 2001), ISBN 0-8493-1222-1
External links [blue-pencil]
-
 Media related to Antioxidants at Wikimedia Commons
Media related to Antioxidants at Wikimedia Commons
Where Does Our Body Obtain the Majority of Its Antioxidants?
Source: https://en.wikipedia.org/wiki/Antioxidant
![{\displaystyle {\ce {{\underset {Oxygen}{O2}}->{\underset {Superoxide}{*O2^{-}}}->[{\ce {Superoxide \atop dismutase}}]{\underset {Hydrogen \atop peroxide}{H2O2}}->[{\ce {Peroxidases \atop catalase}}]{\underset {Water}{H2O}}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/8feecd26377be56b431d182ea9a398ab6b5d3b7f)
Post a Comment for "Where Does Our Body Obtain the Majority of Its Antioxidants?"